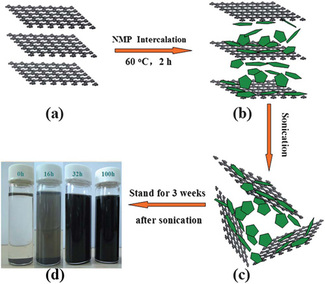
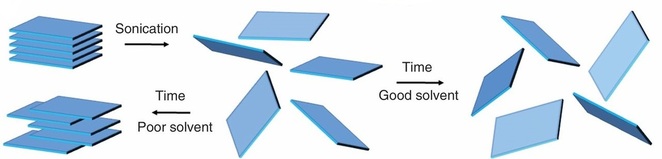
LIQUID EXFOLIATION
Liquid exfoliation makes use of the fact that the individual sheets of the layered materials are held together using van der Waals forces and hence one way of separating the layers is by breaking the Van der Waals forces, which does not seem too hard given that they are weak bonds. By submerging the material in a solvent and shaking it gently in a sonicator, the layers separate easily and the suspension is later centrifuged.
However, if the layers are not separated properly, it is possible for them to interact with each other again to form Van der Waals forces and hence form back the 2D crystal structure. Hence, the key is to find a solvent that has an interaction energy with the layers that is greater than the interaction energy between the layers in the bulk material themselves. When the solvent-layer interaction is stronger than the layer-layer interaction, it is thermodynamically more favourable for the layers to separate and interact with the solvent than stay together.
Another field of research is into the optimal condition of liquid exfoliation or the type of solvent to use to provide the best separation of MoS2 layers. Given the novelty of both the method as well as the focus on Molybdenum disulfide, there are no current common liquids used as solvent in the liquid exfoliation of MoS2. However, some liquids that have shown promising results are:
Sodium cholate hydrate
N-methyl-pyrrolidone
Isopropanol
1-methyl-2-pyrrolidinone (NMP) with a sodium hydroxide (NaOH) assistant.
and the optimal concentration of solvent has been shown to be about 0.3mg/ml.
Sodium cholate hydrate
N-methyl-pyrrolidone
Isopropanol
1-methyl-2-pyrrolidinone (NMP) with a sodium hydroxide (NaOH) assistant.
and the optimal concentration of solvent has been shown to be about 0.3mg/ml.
Advantages
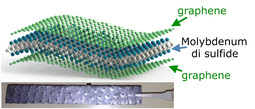
Liquid exfoliation is insensitive to air and water and can be potentially be scaled up to give large quantities of exfoliated material. By playing with the attraction forces between the layers, scientist can also create hybrid materials by adding, for example carbon or graphene in between the MoS2 layers to create new materials with improved properties. Hence, liquid-phase exfoliation methods could lead to a range of thin film applications such as nano-scale hybrids for use in thermoelectrics, supercapacitors or Li-ion batteries. Furthermore, as the electronic properties of TMDs vary relatively slowly with layer number, full exfoliation to monolayers is not necessary and liquid exfoliation is an efficient method if only dispersion to a few-layer flakes is sufficient.
Disadvantages
-Gives dispersion of small nanosheets at low concentrations
Liquid exfoliation is insensitive to air and water and can be potentially be scaled up to give large quantities of exfoliated material. By playing with the attraction forces between the layers, scientist can also create hybrid materials by adding, for example carbon or graphene in between the MoS2 layers to create new materials with improved properties. Hence, liquid-phase exfoliation methods could lead to a range of thin film applications such as nano-scale hybrids for use in thermoelectrics, supercapacitors or Li-ion batteries. Furthermore, as the electronic properties of TMDs vary relatively slowly with layer number, full exfoliation to monolayers is not necessary and liquid exfoliation is an efficient method if only dispersion to a few-layer flakes is sufficient.
Disadvantages
-Gives dispersion of small nanosheets at low concentrations
References:
[1] Nicolosi, V., Chhowalla, M., Kanatzidis, M., Strano, M., & Coleman, J. (2013). Liquid exfoliation of layered materials. Science, 340(6139).
[2] Bang, G., Nam, K., Kim, J., Shin, J., Choi, J., & Choi, S. (2014). Effective liquid-phase exfoliation and sodium ion battery application of MoS2 nanosheets. ACS Applied Materials & Interfaces.
[3]Coleman, J., Lotya, M., O’Neill, A., Bergin, S., King, P., & Khan, U. et al. (2011). Two-dimensional nanosheets produced by liquid exfoliation of layered materials. Science, 331(6017), 568--571.
[4]Pu, F., & others,. (2012). High yield production of inorganic graphene-like materials (MoS₂, WS₂, BN) through liquid exfoliation testing key parameters.
[5] K. F. Mak, C. Lee, J. Hone, J. Shan, T. F. Heinz, Phys. Rev. Lett. 2010, 105, 4.
[6] O’Neill, A., Khan, U., & Coleman, J. (2012). Preparation of high concentration dispersions of exfoliated MoS2 with increased flake size. Chemistry Of Materials, 24(12), 2414--2421.
[7] Newswise.com,. (2014). Engineer Brings New Twist to Sodium-Ion Battery Technology with Discovery of Flexible Molybdenum Disulfide Electrodes. Retrieved 14 September 2014, from http://www.newswise.com/articles/engineer-brings-new-twist-to-sodium-ion-battery-technology-with-discovery-of-flexible-molybdenum-disulfide-electrodes
[1] Nicolosi, V., Chhowalla, M., Kanatzidis, M., Strano, M., & Coleman, J. (2013). Liquid exfoliation of layered materials. Science, 340(6139).
[2] Bang, G., Nam, K., Kim, J., Shin, J., Choi, J., & Choi, S. (2014). Effective liquid-phase exfoliation and sodium ion battery application of MoS2 nanosheets. ACS Applied Materials & Interfaces.
[3]Coleman, J., Lotya, M., O’Neill, A., Bergin, S., King, P., & Khan, U. et al. (2011). Two-dimensional nanosheets produced by liquid exfoliation of layered materials. Science, 331(6017), 568--571.
[4]Pu, F., & others,. (2012). High yield production of inorganic graphene-like materials (MoS₂, WS₂, BN) through liquid exfoliation testing key parameters.
[5] K. F. Mak, C. Lee, J. Hone, J. Shan, T. F. Heinz, Phys. Rev. Lett. 2010, 105, 4.
[6] O’Neill, A., Khan, U., & Coleman, J. (2012). Preparation of high concentration dispersions of exfoliated MoS2 with increased flake size. Chemistry Of Materials, 24(12), 2414--2421.
[7] Newswise.com,. (2014). Engineer Brings New Twist to Sodium-Ion Battery Technology with Discovery of Flexible Molybdenum Disulfide Electrodes. Retrieved 14 September 2014, from http://www.newswise.com/articles/engineer-brings-new-twist-to-sodium-ion-battery-technology-with-discovery-of-flexible-molybdenum-disulfide-electrodes